The Client
GALENICA is one of the largest greek pharmaceutical companies with innovative proprietary medicines that it has already successfully exported to the Balkans. In order to comply with counterfeit drug legislation, it needed a serialization system to use for its medicines production. ZENON AUTOMATION SAICT, in response to this need, designed and implemented a complete serialization in a box system in collaboration with SYSTECH.
The Need
The GALENICA pharmaceutical industry needed an integrated serialization system to monitor and detect prescription drugs throughout the supply chain, thereby achieving compliance with counterfeit drug legislation.
The Solution
Serialization in a box by ZENON AUTOMATION SAICT:
- Easy integration into existing lines
- Automatic product handling and box entry
- Box sorting
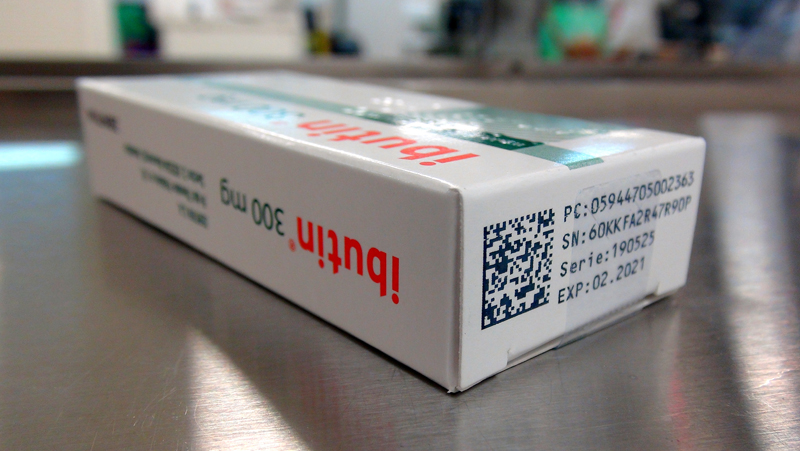
- 2D barcode printing with serialization information
- 2D barcode verification with camera
- Paste tamper labels evident on two sides
- Checking and rejecting non-compliant
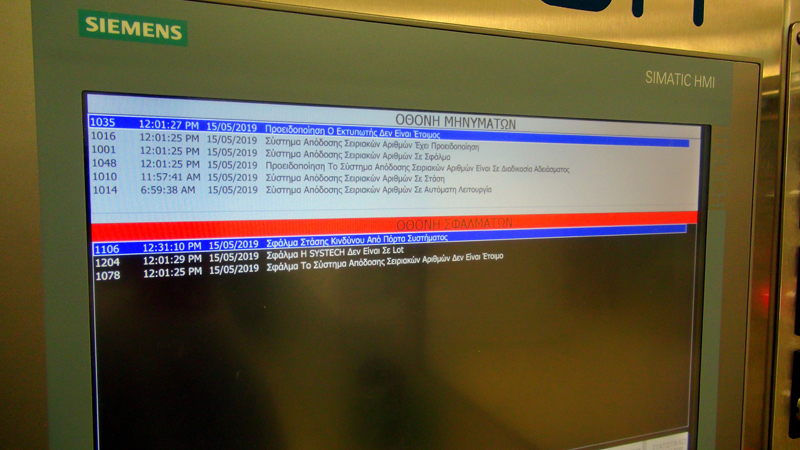
- Screen Error Checking
- Non-compliant code recall
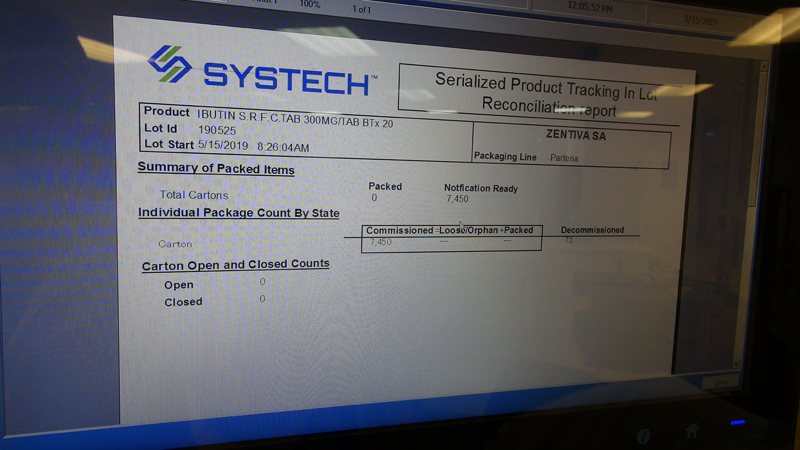
- Integration with SYSTECH serialization software
- Easy and fast change of supplies
The Benefits
- Compliance with serialization legislation
- Integration with existing industrial equipment
- Modern and reliable serialization software
Click on the photographs for a larger view:
If you are interested in a similar solution, please call us on tel. +30 210 6690900 or fill out the contact form below:
Pharmaceuticals / Cosmetics